Abstract
Although infectious agents can act as strong population regulators, knowledge of their spatial distributions in wild Pacific salmon is limited, especially in the marine environment. Characterizing pathogen distributions during early marine residence, a period considered a survival bottleneck for Pacific salmon, may reveal where salmon populations are exposed to potentially detrimental pathogens. Using high-throughput qPCR, we determined the prevalence of 56 infectious agents in 5719 Chinook, 2032 Coho and 4062 Sockeye salmon, sampled between 2008 and 2018, in their first year of marine residence along coastal Western Canada. We identified high prevalence clusters, which often shifted geographically with season, for most of the 41 detected agents. A high density of infection clusters was found in the Salish Sea along the east coast of Vancouver Island, an important migration route and residence area for many salmon populations, some experiencing chronically poor marine survival. Maps for each infectious agent taxa showing clusters across all host species are provided. Our novel documentation of salmon pathogen distributions in the marine environment contributes to the ecological knowledge regarding some lesser known pathogens, identifies salmon populations potentially impacted by specific pathogens, and pinpoints priority locations for future research and remediation.
Introduction
Infectious agents (bacteria, viruses, fungi, and parasites; also referred to as pathogens throughout) are strong regulators of the abundance, distribution, and life histories of animals1. Animal movements and migrations may provide refuge from infections through a departure from infested locations but can also elevate infection risk by exposing hosts to new agents or aggregating stressed hosts2. The interplay of host movements, alternate host ranges, anthropogenic activities, and environmental conditions determines the spatiotemporal distribution of infected hosts, sometimes resulting in clusters of infected individuals known as “hotspots.” For infectious agents that are not well-studied in wild populations or are known to have population level impacts, identifying infection hotspots is an essential step in further understanding or mediating and managing problematic infections. For example, a recent hotspot analysis of the SARS-CoV-2 virus contributed to the epidemiological knowledge of the novel virus while also informing public health resource allocation3.
While many vertebrates experience some level of infection in their lifetimes, not all infections lead to disease and pathogen-induced mortality. However, sub-lethal impacts of infection (e.g. pathogen-induced changes to mobility or behavior) in wild organisms can lead to selective predation, reducing the likelihood of observing infected individuals4,5,6. Mobile aquatic organisms, already elusive, are rarely recovered after perishing in marine or riverine environments, often as a result of predation7,8. In these cases, documenting infectious agent presence independent of, or at least prior to, clinical disease is necessary for determining the distribution of individuals infected by or carrying a given pathogen. Such a task is well-suited to molecular screening methods. High-throughput quantitative polymerase chain reaction (HT-qPCR) platforms can provide datasets ideal for studying dozens of infectious agents simultaneously, across large numbers of test subjects5,9. These methods have revolutionized our ability to study the spatial distribution of infections, including those contributing to indirect mortality via sub-lethal effects, and identify infection hotspots.
Although Pacific salmon (Oncorhynchus spp.) hold great cultural and economic importance, few studies10,11,12 have documented infectious agent distributions (aside from sea lice) in wild salmon over large geographic scales. The majority of relevant knowledge regarding pathogen distributions in the marine environment comes from studies of domesticated fish in marine aquaculture netpens7. Because conditions in netpens differ substantially from the wild in terms of host species, density, predation risk, foraging opportunity, and freedom of movement, infections in aquaculture cannot accurately represent infectious agent distributions in nearby wild populations7,13. Meanwhile, the incidence of marine disease across a broad range of flora and fauna has increased in recent decades14,15,16, highlighting the potential importance of infectious agents for wild salmon during their marine phase. Populations of Sockeye (O. nerka), Coho (O. kisutch), and Chinook (O. tshawytscha) salmon in British Columbia have experienced declines in recent decades17,18,19. One explanation for these declines centers on poor early marine survival20,21,22,23, variously ascribed to predation24, starvation21,25, large-scale oceanic regime shifts26,27, and inter-species competition22,28. A recent study provided evidence that some infectious agents are associated with population-level survival in wild salmon29.
Differences in behavior, physiology and life history between Oncorhynchus spp. of the Northeast Pacific Ocean result in varying exposure and susceptibilty to infectious agents. For example, variations in freshwater rearing habitats have important implications for exposure to pathogens. Sockeye salmon usually rear in lakes for at least a year, whereas Coho salmon typically rear in small streams for a year, and Chinook salmon rear either in large streams for a year or move into the estuary within months of emergence (stream-type vs ocean-type, respectively)30. Freshwater-transmitted infections may carry over into the marine environment, and have measurable impact there11,12,29. Evolved immunological differences between Oncorhynchus spp. result in varied susceptibility to some pathogens31,32, which may manifest in spatial variation in prevalence between species. Migratory behavior in the ocean has important implications for marine infections and our ability to monitor them. Upon ocean entry, most stocks of Sockeye salmon in the Northeast Pacific Ocean migrate rapidly northward33. The majority of Chinook salmon remain within 200 km of their river estuary of origin for their first marine year, except for yearling Columbia River populations that behave more like Sockeye34,35. Coho salmon demonstrate more variability, with some stocks moving shorter distances than others in the first marine year, and larger-bodied individuals migrating further north36.
In this study we sought to identify infection hotspots for 56 infectious agents in Chinook, Sockeye, and Coho salmon during their first marine year. We used a spatial epidemiological tool37 to determine the locations of prevalence clusters for each infectious agent in each salmon host species. We overlaid clusters from all three salmon host species to identify locations in the study area with multiple hotspots across pathogen taxa. We qualitatively related cluster location to known aspects of host population of origin and pathogen biology. Our goal is to provide a resource to further the ecological knowledge regarding these pathogens, inform management activities (e.g., remediation), and prioritize locations for further research on select pathogens.
Results
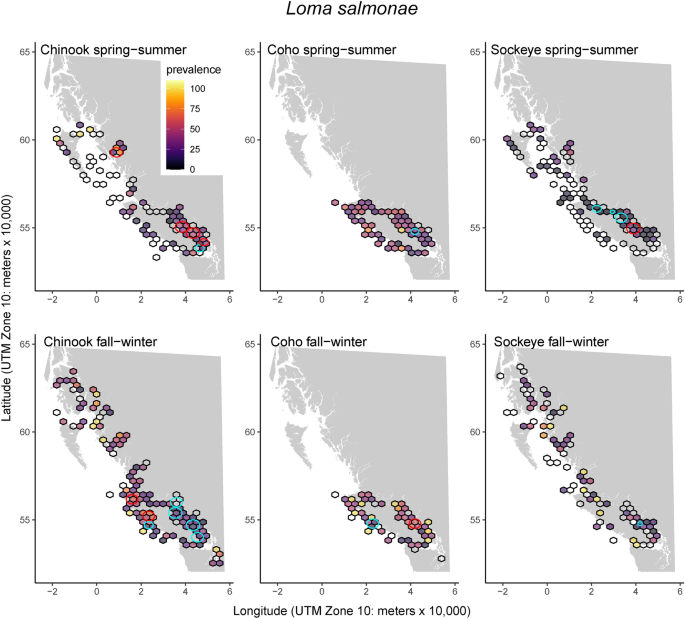
A total of 41 infectious agent taxa were detected (Chinook = 38, Coho = 36, Sockeye = 37) out of a total of 56 assayed (Table 1). Individual fish had positive detections for an average of 4.0 to 4.8 pathogens (total pathogen taxa; range Chinook = 0–12, Coho = 0–10, Sockeye = 0–13), with little variability between species and seasons (Table 1). Infection cluster analysis revealed that most infectious agents had one or more areas with higher or lower infection prevalence than expected by chance. We present the marine-transmitted microsporidian, Loma salmonae, as an illustrative example (Fig. 1). Descriptions of cluster locations for each pathogen and corresponding maps can be found in Supplementary Material 1. Boxplots of pathogen loads divided by species and season are also available in Supplementary Material 1.
Infection clusters for the marine-transmitted microsporidian, Loma salmonae, occurred for all host species but prevalence was greatest in Coho salmon. The color of 30 km hexagons indicates L. salmonae prevalence while empty cells indicate that samples were collected but the pathogen was not detected (0% prevalence). Red circles indicate positive clusters (significantly higher than random likelihood of infection or infection intensity). Blue circles indicate significantly lower than expected regions of infection. Similar maps for 40 other pathogen taxa are available in Supplementary Material 1. Basemap data are from the GSHHG (Global Self-consistent, Hierarchical, High-resolution Geography) Database38. The coordinate system for the data is WGS 1984 and the maps are projected in NAD 1983.
Genetic stock identification
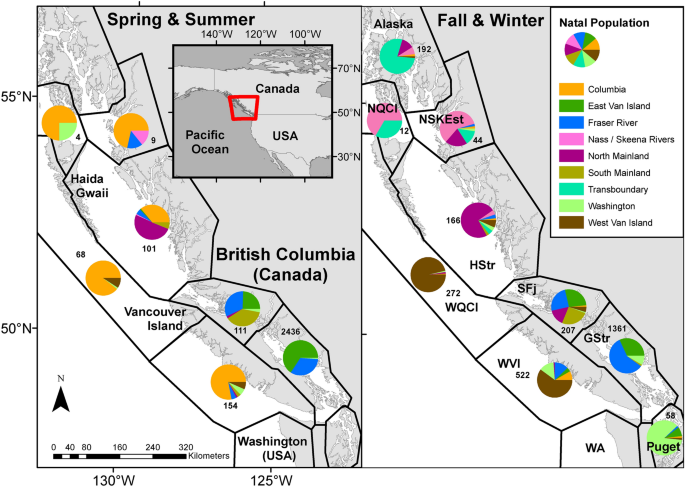
Genetic stock identification revealed that the majority of Chinook and Sockeye salmon sampled along the west coast of Vancouver Island (WVI) in spring-summer originated in the Columbia River (Fig. 2, Tables S1, S3) whereas for Coho salmon, the largest population grouping found in WVI during the spring-summer was from Washington State, followed by the Columbia River (Table S2). In fall-winter, few Columbia River salmon remained in the study area and WVI samples were dominated by WVI origin Chinook and Washington origin Coho salmon. Relatively few Sockeye salmon were sampled in the WVI region during fall-winter. Year round, fish sampled on the east coast of Vancouver Island (EVI) were predominantly from East Vancouver Island rivers and the Fraser River. Chinook salmon sampled in the northern coastal areas tended to originate from northern watersheds whereas for Sockeye salmon, Fraser River origin fish represented the largest population grouping sampled in this region (Fig. 2, Tables S1, S3).
Regional stock compositions, as determined by genetic stock identification, for Chinook salmon collected along the British Columbia coast, 2008–2018. Red outline in inset panel indicates study area extent. Pie charts represent the proportions of stock groupings sampled from regions adapted from DFO’s Marine Adaptive Zones39 (black borders, text in right panel). Total Chinook sampled in each zone provided. Total sampled per zone and stock percentages for all species are in Tables S1, S2, S3. Basemap data are from the GSHHG (Global Self-consistent, Hierarchical, High-resolution Geography) Database38. The coordinate system for the data is WGS 1984 and the maps are projected in NAD 1983.
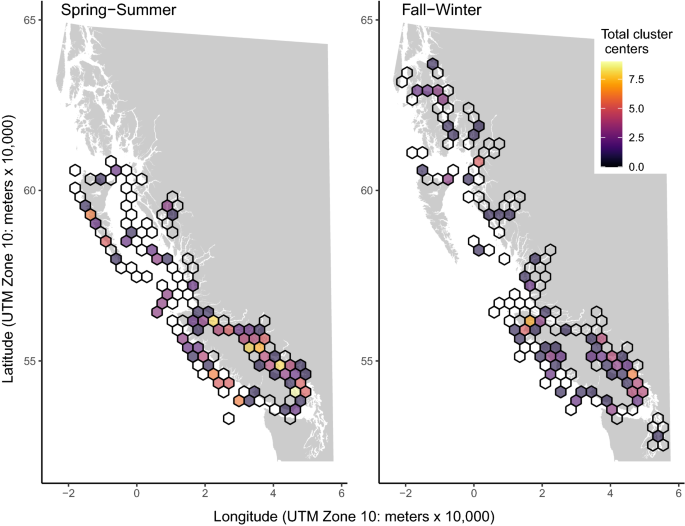
Clusters summed across species and total pathogen taxa
In both seasons, a hexagon containing a high number of cluster centers (nine in spring-summer, six in fall-winter) occurred in southern EVI (Fig. 3, Supplementary Material 2). In spring-summer, this high density hexagon was found in the Gulf Islands, containing the Cowichan River estuary and Salt Spring Island (clusters for Chinook: Piscirickettsia salmonis, Renibacterium salmoninarum, Candidatus Syngnamydia salmonis, Paranucleospora theridion, Myxobolus arcticus, Ichthyophthirius multifiliis, Atlantic Salmon Calicivirus; Sockeye: P. theridion, M. arcticus). During the fall-winter, the most cluster centers fell in a hexagon northwest of the Fraser River estuary, encompassing the mouth of Howe Sound (clusters for Chinook: Rickettsia-like Organism (RLO), Tenacibaculum maritimum, Ceratonova shasta; Sockeye: RLO, M. arcticus, Parvicapsula minibicornis). High density clusters (7–8 cluster centers per 30 km hexagon) were found elsewhere along the EVI in the spring-summer, from the Strait of Georgia (SOG) through Johnstone Strait (Fig. 3, Supplementary Material 2).
Another group of hexagons with high density clusters was found along WVI in the spring-summer (Fig. 3). These clusters were primarily composed of Columbia River salmon (Chinook: 84–100%, Sockeye: 75–100%, Coho: 50–100%) and included C. shasta (n clusters per species: Chinook = 4, Sockeye = 5, Coho = 2), P. minibicornis (n: 4, 0, 0), Piscine orthoreovirus (PRV; n: 3, 2, 0), and Facilispora margolisi (n: 3, 0, 1). WVI was also home to the 30 km hexagon with the highest cluster count in the fall-winter (Fig. 3), a hexagon with seven clusters (five in Chinook, two in Coho) positioned over Quatsino Sound (clusters included F. margolisi, L. salmonae, P. theridion (also in Coho), Parvicapsula kabatai, Tetracapsuloides bryosalmonae, and Erythrocytic Necrosis Virus (ENV, in Coho)).
Visualization of mean total pathogen taxa (the number of pathogen taxa detected per individual) revealed lower counts at the upstream ends of mainland fjords relative to other regions (Supplementary Material 1). Ordinal cluster analysis of total pathogen taxa indicated that all host species had low clusters in the SOG during fall-winter. Coho salmon had consistent (in both spring-summer and fall-winter) positioning of a low cluster in the southern SOG and a high cluster in Quatsino Sound. Chinook had multiple high clusters along the SOG and WVI in spring-summer and a single high cluster centered on Nootka Sound (WVI) in fall-winter. Sockeye had high clusters for total pathogen taxa in Johnstone Strait in spring-summer and a single high cluster along the Alaska coastline in fall-winter.
The total number of statistically significant positive prevalence cluster centers per 30 km hexagon. Empty cells indicate that samples were collected in these locations but no clusters were centered there. An interactive map that identifies pathogen and host species identities for clusters in each hexagon is available in Supplementary Material 2.
Discussion
In an era when many populations of wild Pacific salmon are experiencing consistently poor survival19, and both human activities and rapid climate change are undermining the host–pathogen homeostasis of marine organisms40, greater understanding of the role of infectious agents in the survival of wild salmon is essential. An important step towards this understanding is to determine the distributions of infectious agents within wild salmon hosts in the marine environment. Through the combination of extensive marine sampling, HT-qPCR, and spatial analysis we substantially expanded the current knowledge regarding pathogen distributions in Chinook, Coho, and Sockeye salmon along the coast of British Columbia. Using purpose-built software37 to reveal higher- and lower-than random spatial clustering in pathogen prevalence, we found a high density of clusters for multiple infectious agent taxa within the Salish Sea along the east coast of Vancouver Island, a major migration route and rearing area for multiple species of Pacific salmon. Many notable taxon-specific patterns were also observed and are discussed in Supplementary Material 1.
Locations where hotspots for multiple infectious agents overlap may contribute to detrimental co-infections5,41, can be the result of anthropogenic activities42,43, and could be ideal targets for remediation (e.g., locations where habitat restoration or reductions to municipal or industrial effluent could reduce infection pressure42,44). We found a high density of infection clusters occurring in both seasons in the southern Strait of Georgia. This region is home to the highest urban density in British Columbia, has experienced the most rapid increase in sea surface temperature in the study area45, and is a rearing area and migratory route for multiple declining populations of Pacific salmon18,19.
High densities of pathogen clusters occurred along the east side of Vancouver Island throughout the year. The multiple clusters located around Salt Spring Island at south EVI were composed of freshwater (four) and seawater (five) transmitted pathogens, found in primarily EVI and Fraser River populations of Chinook and Sockeye salmon. Abundant wild and hatchery salmon populations in the Cowichan River, close proximity to human population density42,46, industrial activity, elevated SST45, and poor marine rearing conditions46 are all factors that could have contributed to abundant infection clusters here. Moving north through the SOG, another concentration of infection clusters was detected at the south end of Texada Island, primarily composed of seawater-transmitted infections (e.g., ENV, P. kabatai, I. hoferi), which could be expected following departure from the lower salinity of the Fraser River plume and with increased encounters with Pacific herring, Clupea pallasii (true for ENV and I. hoferi, see below). High densities of infection clusters persisted through the Discovery Islands and Johnstone Strait, regions featuring few feeding opportunities47 and home to a large component of British Columbia’s salmon aquaculture (at the time our samples were collected). Multiple clusters of T. maritimum, a bacterium with evidence of elevated transmission from aquaculture to wild fish in the Discovery Islands48 and potential impacts on wild populations29, were detected in wild Chinook and Sockeye (two clusters for each host) in this region, similar to previous observations49.
Spring-summer infection clusters occurring at a high density along WVI were primarily composed of Columbia River origin salmon. Conditions in the Columbia River including high water temperature, low-current reservoirs, and abundant sediment are ideal for transmission of P. minibicornis and C. shasta50,51, two of the pathogens with multiple clusters along WVI. The migration of juvenile salmon from the Columbia River past Vancouver Island could act as a route of transmission of infectious agents from one region to another. However, C. shasta and P. minibicornis are parasites incapable of horizontal transmission. In contrast, PRV, which also composed spring-summer WVI clusters with predominately Columbia River fish, could be transmitted from one population to another during long-distance migrations52. A recent study found that Columbia River Chinook salmon sampled throughout our study area all shared a common lineage of PRV, suggesting that they were infected in freshwater and then dispersed53. While PRV is already widely dispersed in BC, this potential transmission route could be of future importance in the context of the emergence of virulent pathogens, a phenomenon accelerated by high density fish culture facilities54 such as the many hatcheries on the Columbia River and BC salmon farms.
In the fall-winter period, Quatsino Sound, on the northwest coast of Vancouver Island, was the location with the highest density of infection clusters. Inlets and embayments sometimes host higher infectious agent densities, potentially due to constrained water circulation, elevated temperatures, or seasonal interactions with other host species55,56,57. The Marble River enters Quatsino Sound in an eastern arm and contributes a Chinook salmon population that is resident inside the sound for its first marine year58, and this population composed 78% of our samples here. It is possible, therefore, that unknown factors specific to this population could play a role in the high density of infection clusters here. This region also contains a relatively high density of aquaculture facilities in a narrow fjord, and recent work has shown that nucleic acids of multiple pathogens tend to be elevated around active aquaculture59 and that the probability of infection with some agents increases with proximity to aquaculture48,53.
Fall-winter clusters for PRV in Chinook salmon were detected in Quatsino and Nootka Sounds along WVI, in stocks originating in this area. In addition to the evidence indicating likely transmission of PRV from aquaculture to wild fish53, Atlantic salmon freshwater hatcheries were previously shown to harbor PRV infections60 and thus we might consider whether Pacific salmon enhancement facilities in BC could also play a role in transmission. Further sampling in the WVI region could help determine whether or not a PRV reservoir persists in WVI inlets (either of anthropogenic or natural origin) and could potentially be remeditated.
Cluster locations for total pathogen taxa within individuals did not correspond to locations with overlapping cluster centers from multiple pathogens. This indicates that while clusters from multiple pathogens could be spatially concentrated, individual fish in such a location were not necessarily burdened with a high number of infections. Tucker et al.61 found that mean total pathogen taxa in early marine Chinook salmon increased from 1.5 to 4.2 following marine entrance but then declined to 3.0 in winter. Accordingly, we saw evidence of lower total pathogen taxa within individuals in mainland fjords where collected fish were more likely to have recently left freshwater. While we assayed approximately 10 more pathogens than Tucker et al.61 and we did not see a drop in total pathogen taxa in the fall-winter, the fact that total pathogen taxa tends to average around four in both studies may indicate that there is some upper threshold to the number of different pathogens a Pacific salmon host can sustain, perhaps due to elevated mortality and greater predation risks at higher numbers of co-infections5,6,29. If this is the case, the regions where we observed higher clusters for total pathogen taxa may indicate locations of elevated mortality for each host species (Supplementary Material 1).
Although the majority of infectious agents detected in this study presented similar prevalences and distributions across host salmon species, there were several exceptions. Inter-host-species differences in pathogen prevalence could arise from species-based variation in exposure due to host life histories and food or habitat preferences, species-based variation in susceptibility, statistical artifacts introduced by spatially and seasonally non-random sampling, and potential biases from the use of qPCR (see below). In some cases, the difference in overall prevalence between host species is consistent with what is known regarding host susceptibility, including for P. salmonis62, C. shasta63, M. arcticus64, and Pacific salmon nidovirus (PsNV), Salmon Pescarenavirus 1 (SPAV1), and SPAV265. For L. salmonae, susceptibility is expected to be similar between Chinook and Coho66,67 but we found 1.5–2 times higher prevalence in Coho. Some less-studied taxa, including Aliivibrio salmonicida, Dermocystidium salmonis, Myxobolus insidiosus, and Pacific Salmon Parvovirus (PSPV), presented inter-host-species differences in prevalence that have not previously been described. While unbalanced sampling may have given rise to inter-species variation in these pathogen taxa, our results could also represent epidemiological clues towards aspects of transmission, host susceptibility, and pathogenicity for hosts. For example, the near absence of the freshwater-transmitted PSPV in Chinook and Coho salmon contrasted with high prevalence in Sockeye salmon suggests that Sockeye are considerably more susceptible to infection with this virus. Note that inter-species differences are very apparent for some pathogen taxa when infective load is visualized (Supplementary Material 1).
As is inevitable with an analysis such as ours, a number of simplifying assumptions had to be made. The primary assumption for a spatial analysis of infectious agents in a migratory species is that pathogen profiles in sampled fish represent environmental pathogen presence at the capture location. Clearly this is not an accurate assumption for Sockeye salmon, which are migrating around 10–25 km/day in the marine environment68. Therefore, the reader must consider that in some cases the position of a cluster reflects experiences earlier in the migration. Of all the Pacific salmon species, Chinook might be the most suitable for such an analysis since many stocks tend to remain within 100–200 km of their natal river until their second ocean year, with Columbia River fish being the highly mobile exception34. In our dataset, the sampling locations of 65% of spring-summer and 39% of fall-winter Chinook salmon were less than 50 km from their natal stream (as determined by GSI).
Although our analysis provided a novel, descriptive spatial analysis of dozens of infectious agents in salmon along the British Columbia coast, it could only be conducted as a purely spatial analysis (data from multiple years combined) due to sample size (with the exception of our division of the data into two seasons). This approach assumes that infection clusters are relatively stable from year to year. While this assumption may be valid for a portion of the pathogens we assayed, it is unlikely to be true for all. Pathogens that appeared at lower prevalence, did not have a life cycle requiring an intermediate host, and were more likely to have acute impacts on infected fish (e.g., some bacteria and viruses such as P. rickettsia and Viral Hemorragic Septicemia Virus) might show more inter-annual variation in distribution. Therefore, when interpreting the presence of a given cluster (particularly one for a pathogen with the life history characteristics just described), it is necessary to consider that a high density of infections may have occurred at this location in a subset of or even just one of the study years. Ideally, samples collected evenly over space and time would have allowed us to conduct a spatiotemporal cluster analysis, but this was not logistically possible given limitations imposed by the expense of marine sampling.
Quantitative PCR using TaqMan probes allows for high assay specificity9. While this may be a positive attribute which is responsible for the relatively low false positive rate of PCR, it may also enable us to overlook closely-related but divergent lineages of certain pathogens, particularly for agents with high mutation rates, most notably RNA viruses69. Consider the example of SPAV1 and SPAV2. Had we only designed primers for one of these closely-related viruses, we would have hypothesized that a salmon arenavirus is found in solely Chinook or Sockeye salmon. Therefore, we must consider that in cases where we saw high host tropism (e.g., PSPV) there may have been a lineage capable of infecting other hosts that went undetected due to the specificity of our assays.
Our study represents the most comprehensive assessment of the marine distributions of infectious agents in three ecologically, culturally, and commercially important salmon species. We have provided observations of the marine distribution of dozens of pathogen taxa for which little is currently known in wild Pacific salmon. We anticipate that these distributions will prove useful to those conducting further studies of pathogens in Pacific salmon as well as those seeking to identify potential locations for remediation. Our high-throughput qPCR methodology, especially paired with new innovations in sampling environmental DNA70, is an efficient tool for monitoring a broad range of salmon pathogens and would prove quite useful for the proactive identification of pathogens in aquaculture and hatchery environments. For research, an important next step is to identify the intrinsic (fish related) and extrinsic (environmental) factors associated with the distributions of pathogens that we have described in this study.
Methods
Field sampling
Data used in this study were from a database created over the course of the Strategic Salmon Health Initiative https://www.canada.ca/en/fisheries-oceans/news/2016/05/strategic-salmon-health-initiative.html and subsets of these data have been used previously in unrelated analyses29,49,71,72. Pacific salmon (n: Chinook = 5719, Coho = 2059, Sockeye = 3387) in their first year of ocean residence (April through March of the following year) were opportunistically collected during several Fisheries and Oceans Canada research programs from 2008 to 2018 (scientific fishing permit MECTS # 2014-502-00249). Samples were collected as far north as Fredrick Sound, Alaska (56\(^{\circ }\)N) and as far south as the mid-Puget Sound, Washington (47.5\(^{\circ }\)N, Fig. 2). Methods used to capture salmon included mid-water trawl (n: Chinook = 4308, Coho = 1869, Sockeye = 2431), purse seine (n: Chinook = 458, Coho = 87, Sockeye = 956), and beach seine (n: Chinook = 953, Coho = 103, Sockeye = 0). Fish were either dead upon landing or euthanized in buffered tricaine methanesulfonate. A tissue sample was taken from the adipose fin or operculum and preserved in 95% ethanol to determine the population of origin for each fish, a process known as genetic stock identification (GSI)73. Fish were either dissected in the field or frozen at − 80 °C after capture and subsequently dissected in the lab. In the field, tissues samples including gill, brain, liver, head kidney, and heart were placed in RNAlater (Qiagen, MD, USA) for 24 h at 4 °C and then frozen at − 80 °C.
All methods were performed in accordance with the relevant guidelines and regulations according to the Canadian Council on Animal Care’s (CCAC) Guide to the Care and Use of Experimental Animals, and project protocols were approved by the federal department of Fisheries and Oceans Canada (DFO) through its Pacific Region Animal Care Committee (Animal Use Protocol Number: 13-008). Where applicable, this study is reported in accordance with ARRIVE guidelines. Because collection of samples was reliant on the operations of multi-use research vessels and the study was observational in nature so that no experimental manipulations were imposed, not all ARRIVE guidelines are applicable.
Because sample distributions, population composition, diets, and infectious agent profiles for salmon have been shown to vary in a biologically significant manner within the first marine year11,61,74, we split samples into the “spring-summer” (April through August, n: Chinook = 2883, Coho = 1062, Sockeye = 2826) and “fall-winter” (September through March, n: Chinook = 2836, Coho = 997, Sockeye = 561) sample periods (sample size precluded further seasonal division). We utilized this same seasonal division in a recent study of the associations between infectious agent prevalence and population-level survival29. No samples were excluded from analysis.
Molecular methods
Tissue samples were screened for the presence of 56 infectious agent taxa (Tables 1, S4, Supplementary Material 1), using HT-qPCR on the Fluidigm Biomark Dynamic ArrayTM microfluidics platform (Fluidigm, San Francisco, CA, USA) at the Pacific Biological Station, Nanaimo, British Columbia, Canada. This platform has been analytically validated for quantitative infectious agent profiling in salmon tissue9 and applied to dozens of studies in Pacific and Atlantic salmon71,75,76. Infectious agent taxa were chosen based on knowledge of their presence in Canada, evidence of their association with disease worldwide, or recent discovery by our group9,65,77. Assays utilizing Taqman probes (Table S4, Supplementary Material 1) were designed to target both RNA and DNA. Not all of the same assays were used over the course of the qPCR runs (over 200 dynamic arrays run over the course of 6 years), as some new assays were developed and others were removed after no detections across previous studies (% of population assayed per pathogen reported in Table 1). Detailed laboratory methods are provided in Supplementary Material 1. We also determined “total pathogen taxa” per individual fish by tallying the number of detected pathogens found in each individual.
Statistical analyses
We used resampling simulations (SatScan [https://www.satscan.org]) to identify clusters of high (and low) prevalence for each infectious agent that was detected at any prevalence greater than zero (Table 1). The resampling simulations identify areas of higher or lower prevalence than would be expected if the pathogen was distributed uniformly across space78. Satscan places circular “scanning windows” of varying sizes around pre-defined geographic coordinates to determine, for a given infectious agent, whether the group of samples within a given window are significantly different in prevalence (percent positive) from all samples outside the scanning window. Statistical significance was determined using a likelihood ratio test to compare groups within and outside the scanning window, and an accompanying p-value generated through Monte Carlo simulation wherein the dataset outside the scanning window is randomly resampled 1000 times (clusters with \(p < 0.05\) are presented in maps). All analyses were conducted in the R statistical language79 using the rsatscan package80. Example R code is provided in Supplementary Material 1.
Samples from all years were aggregated across an evenly spaced grid of hexagons (10 km from center to center) and scanning windows of various sizes were applied to hexagon centers. Thus, this was a purely spatial analysis with the only temporal aspect being that a separate analysis was performed for the spring-summer and fall-winter seasons for each infectious agent. By conducting a set of year-specific (but otherwise identical) analyses on a subset of pathogens, we determined that a purely spatial analysis adequately (if not conservatively) represented cluster locations identified in the year-specific analysis, without creating additional, spurious clusters. We elected to use the purely spatial approach due to its demonstrated ability to identify year-specific clusters, the sparsity of data in some years, and the extreme number of analyses and figures that a year-specific analysis would entail. Because we wanted to prevent scanning windows from crossing land masses (particularly Vancouver Island) we set the maximum scanning window radius to 30 km, with the assumption that the inference from a single large cluster (as might occur if we used the default window size limit, a window that encompasses 50% of the total population sample size) would be similar to that from several adjacent clusters. For a given infectious agent, significant clusters were not allowed to overlap. For all infectious agents we set the model type to the Bernoulli distribution78, which simply requires the input of geographic locations and the number of positive and negative detections per location. We also performed a similar cluster analysis on total pathogen taxa (a tally of the pathogens detected per individual) but using the ordinal model type81. To visualize the spatial distribution of clusters we calculated and color-coded infectious agent prevalence for a grid of 30 km hexagons superimposed across the sample locations. Significant (\(p < 0.05\)) scanning windows of both high (red) and low (blue) clusters were superimposed onto this base map. We also tallied cluster centers across all infectious agents and host species using the 30 km hexagonal grid to visually determine whether there were any areas with high densities of clusters across multiple pathogen taxa.
Data availibility
All data generated or analysed during this study are included in this published article (and its Supplementary Information files).
References
-
May, R. M. Parasitic infections as regulators of animal populations: The dynamic relationship between parasites and their host populations offers clues to the etiology and control of infectious disease. Am. Sci. 71, 36–45 (1983).
-
Altizer, S., Bartel, R. & Han, B. A. Animal migration and infectious disease risk. Science 331, 296–302. https://doi.org/10.1126/science.1194694 (2011).
-
Hohl, A., Delmelle, E. M., Desjardins, M. R. & Lan, Y. Daily surveillance of COVID-19 using the prospective space-time scan statistic in the United States. Spatial Spatio-temporal Epidemiol. 34, 100354 (2020).
-
Mesa, M. G., Poe, T. P., Maule, A. G. & Schreck, C. B. Vulnerability to predation and physiological stress responses in juvenile chinook salmon (Oncorhynchus tshawytscha) experimentally infected with Renibacterium salmoninarum. Can. J. Fish. Aquat. Sci. 55, 1599–1606 (1998).
-
Miller, K. M. et al. Infectious disease, shifting climates, and opportunistic predators: Cumulative factors potentially impacting wild salmon declines. Evol. Appl. 7, 812–855 (2014).
-
Furey, N. B. et al. Infected juvenile salmon can experience increased predation during freshwater migration. R. Soc. Open Sci. 8, 201522 (2021).
-
Bakke, T. A. & Harris, P. D. Diseases and parasites in wild Atlantic salmon (Salmo salar) populations. Can. J. Fish. Aquat. Sci. 55, 247–266 (1998).
-
Patterson, D., Skibo, K., Barnes, D., Hills, J. & Macdonald, J. The influence of water temperature on time to surface for adult sockeye salmon carcasses and the limitations in estimating salmon carcasses in the Fraser River, British Columbia. N. Am. J. Fish. Manag. 27, 878–884 (2007).
-
Miller, K. M. et al. Report on the performance evaluation of the Fluidigm BioMark platform for high-throughput microbe monitoring in salmon. DFO Can. Sci. Advis. Sec. Res. Doc. 2016/038. xi + 282 p. (2016).
-
Arkoosh, M. R. et al. Survey of pathogens in juvenile salmon Oncorhynchus spp. migrating through pacific northwest estuaries. J. Aquat. Anim. Health 16, 186–196. https://doi.org/10.1577/H03-071.1 (2004).
-
Jacobson, K. C., Teel, D., Van Doornik, D. M. & Casillas, E. Parasite-associated mortality of juvenile Pacific salmon caused by the trematode Nanophyetus salmincola during early marine residence. Mar. Ecol. Prog. Ser. 354, 235–244 (2008).
-
Sandell, T., Teel, D. J., Fisher, J., Beckman, B. & Jacobson, K. Infections by Renibacterium salmoninarum and Nanophyetus salmincola (Chapin) are associated with reduced growth of juvenile Chinook salmon, Oncorhynchus tshawytscha (Walbaum), in the Northeast Pacific Ocean. J. Fish Dis. 38, 365–378 (2015).
-
Laurin, E. et al. Histopathological and novel high-throughput molecular monitoring data from farmed salmon (Salmo salar and Oncorhynchus spp.) in British Columbia, Canada, from 2011–2013. Aquaculture 499, 220–234 (2019).
-
Harvell, C. et al. Emerging marine diseases-climate links and anthropogenic factors. Science 285, 1505–1510 (1999).
-
Adlard, R. D., Miller, T. L. & Smit, N. J. The butterfly effect: Parasite diversity, environment, and emerging disease in aquatic wildlife. Trends Parasitol. 31, 160–166 (2015).
-
Tracy, A. M., Pielmeier, M. L., Yoshioka, R. M., Heron, S. F. & Harvell, C. D. Increases and decreases in marine disease reports in an era of global change. Proc. R. Soc. B 286, 20191718 (2019).
-
Peterman, R. M. & Dorner, B. A widespread decrease in productivity of sockeye salmon (Oncorhynchus nerka) populations in western North America. Can. J. Fish. Aquat. Sci. 69, 1255–1260 (2012).
-
Zimmerman, M. S. et al. Spatial and temporal patterns in smolt survival of wild and hatchery coho salmon in the Salish Sea. Mar. Coast. Fish. 7, 116–134 (2015).
-
COSEWIC. Cosewic assessment and status report on the chinook salmon Oncorhynchus tshawytscha, designatable units in southern British Columbia (part one—designatable units with no or low levels of artificial releases in the last 12 years), in Canada. Tech. Rep., Committee on the Status of Endangered Wildlife in Canada. Ottawa (2019).
-
Peterman, R. M., Pyper, B. J., Lapointe, M. F., Adkison, M. D. & Walters, C. J. Patterns of covariation in survival rates of British Columbian and Alaskan sockeye salmon (Oncorhynchus nerka) stocks. Can. J. Fish. Aquat. Sci. 55, 2503–2517 (1998).
-
Beamish, R. J. & Mahnken, C. A critical size and period hypothesis to explain natural regulation of salmon abundance and the linkage to climate and climate change. Prog. Oceanogr. 49, 423–437 (2001).
-
Cunningham, C. J., Westley, P. A. & Adkison, M. D. Signals of large scale climate drivers, hatchery enhancement, and marine factors in Yukon River Chinook salmon survival revealed with a Bayesian life history model. Glob. Change Biol. 24, 4399–4416 (2018).
-
Claiborne, A. M. et al. Correspondence between scale growth, feeding conditions, and survival of adult chinook salmon returning to the southern salish sea: Implications for forecasting. Prog. Oceanogr. 198, 102443 (2020).
-
Nelson, B. W., Walters, C. J., Trites, A. W. & McAllister, M. K. Wild Chinook salmon productivity is negatively related to seal density and not related to hatchery releases in the Pacific Northwest. Can. J. Fish. Aquat. Sci. 76, 447–462 (2018).
-
Duffy, E. J. & Beauchamp, D. A. Rapid growth in the early marine period improves the marine survival of Chinook salmon (Oncorhynchus tshawytscha) in Puget Sound, Washington. Can. J. Fish. Aquat. Sci. 68, 232–240 (2011).
-
Ohlberger, J., Scheuerell, M. D. & Schindler, D. E. Population coherence and environmental impacts across spatial scales: A case study of Chinook salmon. Ecosphere 7, e01333 (2016).
-
Daly, E. A., Brodeur, R. D. & Auth, T. D. Anomalous ocean conditions in 2015: Impacts on spring Chinook salmon and their prey field. Mar. Ecol. Prog. Ser. 566, 169–182 (2017).
-
Ruggerone, G. T. & Connors, B. M. Productivity and life history of sockeye salmon in relation to competition with pink and sockeye salmon in the North Pacific Ocean. Can. J. Fish. Aquat. Sci. 72, 818–833 (2015).
-
Bass, A. L. et al. Identification of infectious agents in early marine chinook and coho salmon associated with cohort survival. (in Review) (2022).
-
Groot, G. Pacific Salmon Life Histories (UBC Press, 1991).
-
Ramsay, J., Speare, D. J., Dawe, S. & Kent, M. Xenoma formation during microsporidial gill disease of salmonids caused by Loma salmonae is affected by host species (Oncorhynchus tshawytscha, O. kisutch, O. mykiss) but not by salinity. Dis. Aquat. Org. 48, 125–131 (2002).
-
Lapatra, S. E. Factors affecting pathogenicity of infectious hematopoietic necrosis virus (IHNV) for salmonid fish. J. Aquat. Anim. Health 10, 121–131 (1998).
-
Tucker, S. et al. Seasonal stock-specific migrations of juvenile sockeye salmon along the west coast of North America: Implications for growth. Trans. Am. Fish. Soc. 138, 1458–1480 (2009).
-
Trudel, M. et al. Distribution and migration of juvenile Chinook salmon derived from coded wire tag recoveries along the continental shelf of western North America. Trans. Am. Fish. Soc. 138, 1369–1391 (2009).
-
Riddell, B. E. et al. Ocean ecology of chinook salmon. Ocean Ecol. Pacific Salmon Trout 555–696 (2018).
-
Beacham, T. D. et al. Stock-specific size and migration of juvenile coho salmon in British Columbia and Southeast Alaska waters. Mar. Coast. Fish. 8, 292–314 (2016).
-
Kulldorff, M. Satscantm user guide (2006).
-
Wessel, P. & Smith, W. H. A global, self-consistent, hierarchical, high-resolution shoreline database. J. Geophys. Res. Solid Earth 101, 8741–8743 (1996).
-
DFO. Framework for characterizing Conservation Units of Pacific salmon (Oncorhynchus spp.) for implementing the Wild Salmon Policy. (2009).
-
Burge, C. A. & Hershberger, P. K. Climate change can drive marine diseases. In Marine Disease Ecology (eds Behringer, D. C. et al.) 83–97 (Oxford University Press, 2020).
-
Kotob, M. H., Menanteau-Ledouble, S., Kumar, G., Abdelzaher, M. & El-Matbouli, M. The impact of co-infections on fish: A review. Vet. Res. 47, 98 (2017).
-
Bojko, J., Lipp, E., Ford, A. & Behringer, D. Pollution can drive marine diseases. In Marine Disease Ecology 95–113 (Oxford University Press, 2020).
-
Behringer, D. C., Wood, C. L., Krkošek, M. & Bushek, D. Disease in fisheries and aquaculture. In Marine Disease Ecology (eds Behringer, D. C. et al.) 183–213 (Oxford University Press, 2020).
-
Lamb, J. B. et al. Seagrass ecosystems reduce exposure to bacterial pathogens of humans, fishes, and invertebrates. Science 355, 731–733 (2017).
-
Amos, C. L., Martino, S., Sutherland, T. & Al Rashidi, T. Sea surface temperature trends in the coastal zone of British Columbia, Canada. J. Coast. Res. 31, 434–446 (2015).
-
Chittenden, C. et al. Estuarine and marine diets of out-migrating chinook salmon smolts in relation to local zooplankton populations, including harmful blooms. Estuarine Coast. Shelf Sci. 200, 335–348 (2018).
-
Journey, M., Trudel, M., Young, G. & Beckman, B. Evidence for depressed growth of juvenile Pacific salmon (Oncorhynchus) in Johnstone and Queen Charlotte Straits, British Columbia. Fish. Oceanogr. 27, 174–183 (2018).
-
Bateman, A. W. et al. Atlantic salmon farms are a likely source of Tenacibaculum maritimum infection in migratory Fraser River sockeye salmon. Can. J. Fish. Aquat. Sci. 79, 1225–1240 (2022).
-
Nekouei, O. et al. Detection and assessment of the distribution of infectious agents in juvenile Fraser River Sockeye Salmon, Canada, in 2012 and 2013. Front. Microbiol. 9, 3221 (2018).
-
Stocking, R. W. & Bartholomew, J. L. Distribution and habitat characteristics of Manayunkia speciosa and infection prevalence with the parasite Ceratomyxa shasta in the Klamath River, Oregon-California. J. Parasitol. 93, 78–88 (2007).
-
Ray, R. A. & Bartholomew, J. L. Estimation of transmission dynamics of the Ceratomyxa shasta actinospore to the salmonid host. Parasitology 140, 907–916 (2013).
-
Garseth, Å. H., Ekrem, T. & Biering, E. Phylogenetic evidence of long distance dispersal and transmission of piscine reovirus (PRV) between farmed and wild Atlantic salmon. PLoS ONE 8, e82202 (2013).
-
Mordecai, G. J. et al. Aquaculture mediates global transmission of a viral pathogen to wild salmon. Sci. Adv. 7, eabe2592 (2021).
-
Pulkkinen, K. et al. Intensive fish farming and the evolution of pathogen virulence: The case of columnaris disease in Finland. Proc. R. Soc. B Biol. Sci. 277, 593–600 (2010).
-
Johnson, S., Blaylock, R., Elphick, J. & Hyatt, K. Disease induced by the sea louse (Lepeophteirus salmonis) (Copepoda: Caligidae) in wild sockeye salmon (Oncorhynchus nerka) stocks of Alberni Inlet, British Columbia. Can. J. Fish. Aquat. Sci. 53, 2888–2897 (1996).
-
Rhodes, L. D., Durkin, C., Nance, S. L. & Rice, C. A. Prevalence and analysis of Renibacterium salmoninarum infection among juvenile Chinook salmon Oncorhynchus tshawytscha in North Puget Sound. Dis. Aquat. Org. 71, 179–190 (2006).
-
Hershberger, P. et al. Prevalence of viral erythrocytic necrosis in Pacific herring and epizootics in Skagit Bay, Puget Sound, Washington. J. Aquat. Anim. Health 21, 1–7 (2009).
-
Trudel, M. et al. Estimating winter mortality in juvenile Marble River Chinook salmon. N. Pac. Anadr. Fish Comm. Doc 1426, 14 (2012).
-
Shea, D. et al. Environmental DNA from multiple pathogens is elevated near active Atlantic salmon farms. Proc. R. Soc. B 287, 20202010 (2020).
-
Bateman, A. W. et al. Descriptive multi-agent epidemiology via molecular screening on Atlantic salmon farms in the northeast Pacific Ocean. Sci. Rep. 11, 1–15 (2021).
-
Tucker, S., Li, S., Kaukinen, K. H., Patterson, D. A. & Miller, K. M. Distinct seasonal infectious agent profiles in life-history variants of juvenile Fraser River Chinook salmon: An application of high-throughput genomic screening. PLoS ONE 13, e0195472 (2018).
-
Jones, S. R. Characterization of Piscirickettsia salmonis and salmonid rickettsial septicaemia to inform pathogen transfer risk assessments in British Columbia. DFO Canadian Science Advisory Secretariat Research Document (2019).
-
Zinn, J., Johnson, K., Sanders, J. & Fryer, J. Susceptibility of salmonid species and hatchery strains of Chinook salmon (Oncorhynchus tshawytscha) to infections by Ceratomyxa shasta. J. Fish. Board Canada 34, 933–936 (1977).
-
Kent, M., Margolis, L., Whitaker, D., Hoskins, G. & McDonald, T. Review of Myxosporea of importance in salmonid fisheries and aquaculture in British Columbia. Folia Parasitol. 41, 27–38 (1994).
-
Mordecai, G. J. et al. Endangered wild salmon infected by newly discovered viruses. eLife 8, e47615 (2019).
-
Becker, J. A. & Speare, D. J. Transmission of the microsporidian gill parasite, loma salmonae. Anim. Health Res. Rev. 8, 59–68 (2007).
-
Speare, D. J. & Lovy, J. Loma salmonae and related species. In Fish Parasites: Pathobiology and Protection, (eds Woo, P. T. & Buchmann, K.) chap. 7, 109 (CABI, 2012).
-
Clark, T. D. et al. Tracking wild sockeye salmon smolts to the ocean reveals distinct regions of nocturnal movement and high mortality. Ecol. Appl. 26, 959–978 (2016).
-
Duffy, S. Why are RNA virus mutation rates so damn high?. PLoS Biol. 16, e3000003 (2018).
-
Deeg, C. M. et al. Environmental DNA survey of the winter Salmonosphere in the Gulf of Alaska. N. Pac. Anadr. Fish Comm. Tech. Rep. 17, 116–120 (2021).
-
Thakur, K. K. et al. A comparison of infectious agents between hatchery-enhanced and wild out-migrating juvenile chinook salmon (Oncorhynchus tshawytscha) from Cowichan River, British Columbia. FACETS 3, 695–721 (2018).
-
Nekouei, O. et al. Comparison of infectious agents detected from hatchery and wild juvenile coho salmon in British Columbia, 2008–2018. PLoS ONE 14, e0221956 (2019).
-
Beacham, T. D. et al. Estimation of stock composition and individual identification of chinook salmon across the pacific rim by use of microsatellite variation. Trans. Am. Fish. Soc. 135, 861–888 (2006).
-
Duffy, E. J., Beauchamp, D. A., Sweeting, R. M., Beamish, R. J. & Brennan, J. S. Ontogenetic diet shifts of juvenile Chinook salmon in nearshore and offshore habitats of Puget Sound. Trans. Am. Fish. Soc. 139, 803–823 (2010).
-
Di Cicco, E. et al. Heart and skeletal muscle inflammation (HSMI) disease diagnosed on a British Columbia salmon farm through a longitudinal farm study. PLoS ONE 12, e0171471 (2017).
-
Miller, K. M., Günther, O. P., Li, S., Kaukinen, K. H. & Ming, T. J. Molecular indices of viral disease development in wild migrating salmon. Conserv. Physiol. 5, cox036 (2017).
-
Mordecai, G. J. et al. Discovery and surveillance of viruses from salmon in British Columbia using viral immune-response biomarkers, metatranscriptomics, and high-throughput RT-PCR. Virus Evol. 7, veaa069 (2021).
-
Kulldorff, M. & Nagarwalla, N. Spatial disease clusters: Detection and inference. Stat. Med. 14, 799–810 (1995).
-
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2019).
-
Kleinman, K. rsatscan: Tools, Classes, and Methods for Interfacing with SaTScan Stand-Alone Software. R package version 0.3.9200 (2015).
-
Jung, I., Kulldorff, M. & Klassen, A. C. A spatial scan statistic for ordinal data. Stat. Med. 26, 1594–1607 (2007).
Acknowledgements
Thanks to Brian Riddell, co-lead of SSHI; Amy Tabata for SSHI database management; co-op students, field technicians, and others who conducted sampling across many different projects including; Marc Trudel, Chrys Neville, Tyler Zubkowski, Yeongha Jung, Kelsey Flynn, Kim Jonsen, Collin Wallace, Stephen Yu, Anna Yao, Claire Rycroft, Devan Johnson, and Dylan Conover. Thanks to Gideon Mordecai as well as two anonymous reviewers for suggested revisions. This research was part of the Strategic Salmon Health Initiative, co-led by KMM and co-funded by the Pacific Salmon Foundation (Salish Sea Marine Survival Program), Genome British Columbia, Fisheries and Oceans Canada, and the Canadian Coast Guard. AB was supported by a Mitacs Fellowship.
Author information
Authors and Affiliations
Contributions
K.M.M. conceived the study and K.M.M. and S.G.H. supervised laboratory and statistical analyses. K.H.K., S.L., and T.M. conducted laboratory analyses. A.L.B. and A.W.B. conducted statistical analyses. D.A.P. acquired samples for analysis. All authors made substantial contributions to writing and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bass, A.L., Bateman, A.W., Kaukinen, K.H. et al. The spatial distribution of infectious agents in wild Pacific salmon along the British Columbia coast. Sci Rep 13, 5473 (2023). https://ift.tt/qFn84TH
-
Received:
-
Accepted:
-
Published:
-
DOI: https://ift.tt/qFn84TH
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.
The spatial distribution of infectious agents in wild Pacific salmon along the British Columbia coast | Scientific Reports - Nature.com
Read More

No comments:
Post a Comment